I. What are the Enzymes and Processes of Glycolysis?
We again cannot stress the apologies we have to give for the amount of enzymes and steps involved in memorizing this process. There’s a lot more enzymes we’ll encounter in this section but let’s first get our feet a little wet.
It’s definitely daunting not knowing which enzymes and steps to focus on! Though as the saying goes “all is fair game”, there is definitely a hierarchy in terms of what to focus on!
In the article, we’ll focus on the high yield material you’ll need to know for the MCAT: then, we’ll slowly start to chip away at the more rarer topics. Try this approach in other subjects to make your MCAT prep more efficient.
II. Glycolytic Pathway
Let's start off nice and general with an overview of glycolysis! From here we’ll get into the finer details of the pathway!
A. Net Molecular and Energetic Results of Glycolysis
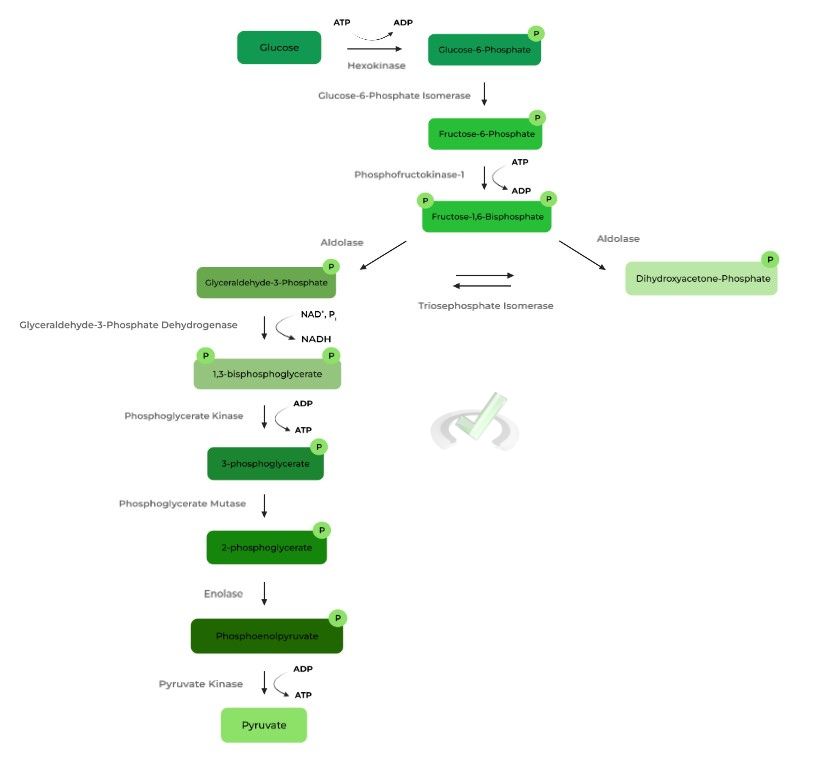
Glycolysis results in the oxidation of 1 glucose into 2 pyruvate molecules while on the way producing 2 net ATPs from substrate level phosphorylation and 2 NADH molecules.
B. Irreversible Enzymes/Steps
As implied by their name, these enzymes and steps cannot undergo the reverse reaction from the ones they catalyze. The 3 irreversible glycolytic enzymes are 1) hexokinase/glucokinase, 2) phosphofructokinase-1, and 3) pyruvate kinase.
I. Glucose to Glucose-6-Phosphate (Hexokinase/Glucokinase)
The first step catalyzes the addition of a phosphate group on carbon 6 of a glucose molecule. This works to “trap” the glucose in the cell as the GLUT transporters are specific for a non phosphorylated glucose.
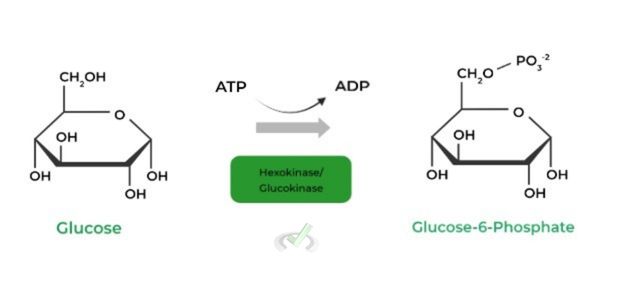
G6P also cannot diffuse out via the cell membrane due to the added charge from the phosphate group, preventing lipid core interaction.
While hexokinase is more prominent in the body, especially the skeletal muscle, glucokinase, another isoform, lies primarily in hepatocytes & pancreatic 𝛽-islets cells.
II. Fructose-6-Phosphate to Fructose-1,6-Bisphosphate (Phosphofructokinase-1)
This is the rate-limiting step in glycolysis and as a result is subject to much allosteric regulation: really look back to negative feedback pathways to understand why!
One example of an allosteric activator of PFK-1 is AMP while some examples of allosteric inhibitors are ATP and citrate.
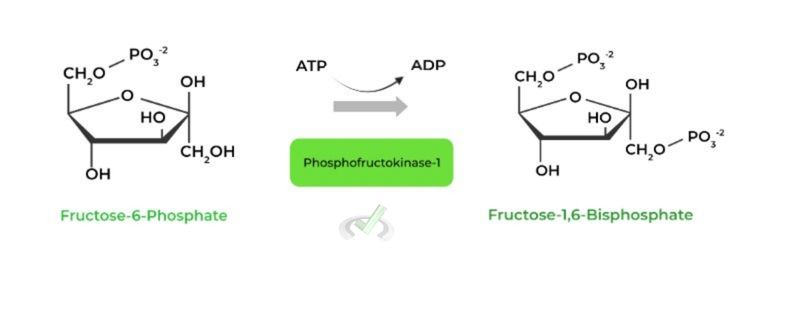
To answer why the certain molecules are activators and inhibitors, remember glycolysis occurs to increase energy production! How should glycolysis be affected when cellular energy levels are low (and vice versa?).
III. Phosphoenolpyruvate to Pyruvate (Pyruvate Kinase)
This is the final step of glycolysis and the 2nd substrate level phosphorylation step, the other coming from phosphoglycerate kinase.
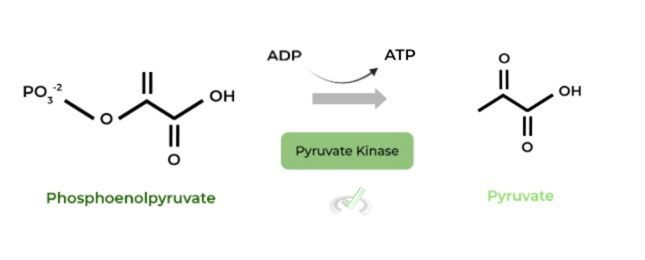
As such, this step produces ATP as well as pyruvate, which can then undergo lactate fermentation or oxidative phosphorylation depending on the oxygen conditions.
C. Other Important Reactions
In addition to the irreversible steps mentioned above, we also want to highlight a few more steps and signify their importance briefly!
I. Dihydroxyacetone-Phosphate to Glyceraldehyde-3-Phosphate (Triosephosphate Isomerase)
As indicated by the name, this is an example of an isomerization reaction, where dihydroxyacetone-phosphate is isomerized to glyceraldehyde-3-phosphate.
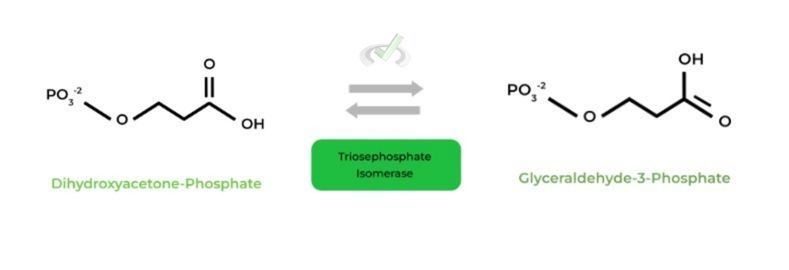
Because DHAP is isomerized to G3P, now 2 DHAPs can undergo steps 6-10, generating a total of 4 ATPS. (Careful though, these aren’t the net ATPs!!)
II. Glyceraldehyde-3-Phosphate to 1,3-Bisphosphoglycerate (Glyceraldehyde-3-Phosphate Dehydrogenase)
Again, as suggested by the enzyme name, this step involves an oxidation-reduction reaction. (Hopefully, you can see a strategy you can use!)
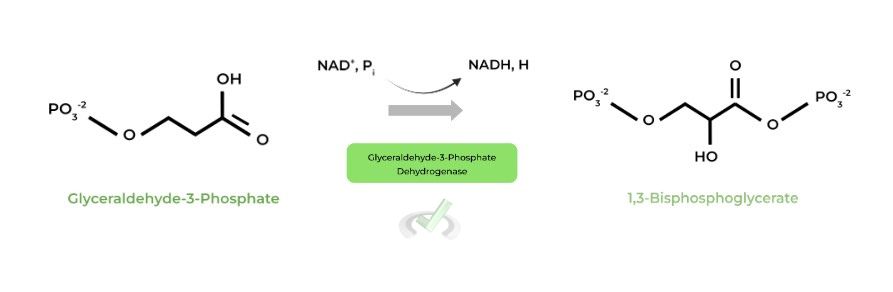
Here, G3P gets oxidized to 1,3-BPG while NAD+ gets reduced to NADH. NADH can then either aid in oxidative phosphorylation or be utilized in lactate fermentation depending on the O2 conditions.
D. Lactate Fermentation
We’ll have a whole other section for oxidative phosphorylation, so we’ll just cover lactate fermentation which occurs in anaerobic (no O2) conditions. The pyruvates are reduced to lactic acid by lactate dehydrogenase, also regenerating NAD+.
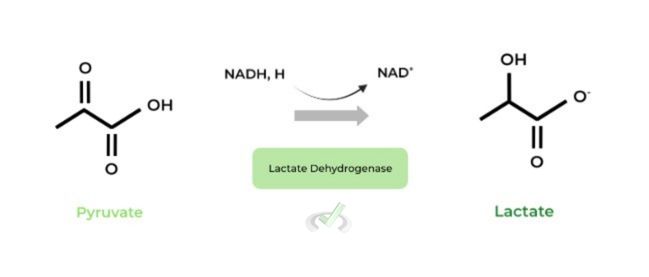
Because the only ATPs generated in anaerobic conditions are in glycolysis, the NAD+ regenerated from lactate fermentation is utilized again for another round of glycolysis!
III. Bridge/Overlap
Why are hexokinase/glucokinase, phosphofructokinase-1, and pyruvate kinase irreversible reactions? We can actually use a little thermodynamics and Gibbs free energy as an explanation!
A. Thermodynamics and Gibbs Free Energy
To give a quick answer, the 3 reactions mentioned above are irreversible due to their large Gibbs free energy values. Let’s go on and take a more detailed look. The rough G values of the steps are shown below:
Compared to the other steps, these have a much larger change in Gibbs free energy! Also note that Gibbs free energy changes are negative, meaning they occur spontaneously.
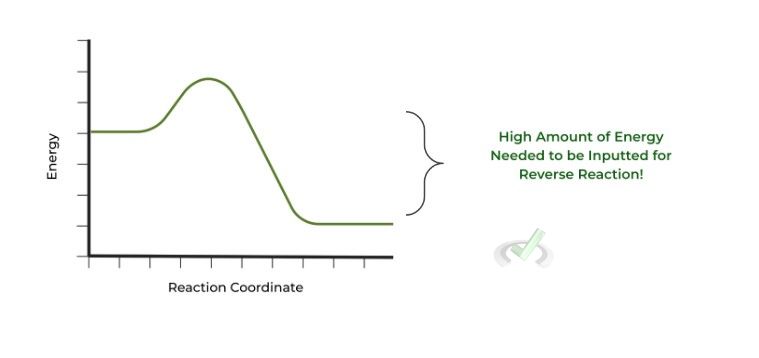
Look at the diagram above! In order for the reverse reaction to occur, a large, positive INPUT of energy must occur in order to power the “uphill” return to the energy state of the reactant!
Because, most likely, there is no large enough energy source available, the reverse reaction WILL NOT occur and is deemed irreversible!
IV. Wrap Up/Key Terms
Let’s take this time to wrap up & concisely summarize what we covered above in the article!
A. Net Molecular and Energetic Results of Glycolysis
Glycolysis is the 10 step process that results in the oxidation of 1 glucose into 2 pyruvates, also producing NADH and ATP in the process.
B. Irreversible Enzymes/Steps
There are 3 irreversible enzyme steps with glycolysis due to their large, negative change in their Gibbs free energy: 1) hexokinase/glucokinase, 2) phosphofructokinase-1, and 3) pyruvate kinase.
I. Glucose to Glucose-6-Phosphate (Hexokinase/Glucokinase)
Occurs in order to “trap”glucose in the cell by preventing GLUT protein binding and cell membrane diffusion.
Hexokinase is found widespread throughout the body, especially the skeletal muscles while glucokinase is primarily found in hepatocytes and pancreatic 𝛽-islets cells.
II. Fructose-6-Phosphate to Fructose-1,6-Bisphosphate (Phosphofructokinase-1)
RATE LIMITING step of glycolysis and, as a result, is the target of many allosteric activators and inhibitors.
AMP is an example of an allosteric activator of PFK-1 while ATP and citrate are examples of allosteric inhibitors.
III. Phosphoenolpyruvate to Pyruvate (Pyruvate Kinase)
The last step of glycolysis and the second substrate level phosphorylation reaction. Produces ATP and pyruvates which can enter into oxidative phosphorylation or lactate fermentation depending on O2 conditions.
C. Other Important Reactions
In addition to the above irreversible reactions, there are additional steps in glycolysis that are worth mentioning!
I. Dihydroxyacetone-Phosphate to Glyceraldehyde-3-Phosphate (Triosephosphate Isomerase)
Isomerization reaction which allows for the production of two glyceraldehyde-3-phosphates. These 2 G3Ps can undergo steps 6-10 of glycolysis to produce 4 ATPs (careful again, this is NOT the net ATPs!)
II. Glyceraldehyde-3-Phosphate to 1,3-Bisphosphoglycerate (Glyceraldehyde-3-Phosphate Dehydrogenase)
Oxidation-reduction reaction where G3P is oxidized to 1,3-BGP and NAD+ is reduced to NADH. NADH can then go on to aid in oxidative phosphorylation or regenerate NAD+ in lactic acid fermentation, depending on O2 conditions
D. Lactic Acid Fermentation
In anaerobic conditions (no O2), the only ATPs produced are from glycolysis. In this case, NAD+ must be regenerated in order to undergo another round of glycolysis.
In lactate fermentation, the pyruvates are reduced to lactic acid while NADH is oxidized back into NAD+ in order to undergo another round of glycolysis.


 To help you achieve your goal MCAT score, we take turns hosting these
To help you achieve your goal MCAT score, we take turns hosting these